Unit - 8
Stereochemistry
A three-dimensional (3-D) structure of organic molecules can be represented on paper by using certain conventions. A solid wedge is used to indicate a bond projecting out of the plane of the paper, towards the observer while a dashed wedge is used to indicate a bond projecting out of the plane of the paper, away from the observer.
Organic compounds are broadly divided into two categories
- Acyclic compounds (open chained)
- Cyclic compounds (closed chained)
Acyclic compounds:Acyclic compounds contain open chains of carbon atoms in their molecules. The carbon chains may be either straight chains or branched chains.
CH3-CH2-CH2-CH3 → n-butane
CH3-CH=CH-CH3 → But-2-ene
Cyclic compound:A cyclic compound (ring compound) is a term for a compound in the field of chemistry in which one or more series of atoms in the compound is connected to form a ring. Rings may vary in size from three to many atoms, and include examples where all the atoms are carbon, none of the atoms are carbon (inorganic cyclic compounds), or where both carbon and non-carbon atoms are present (heterocyclic compounds). Depending on the ring size, the bond order of the individual links between ring atoms, and their arrangements within the rings, carbocyclic and heterocyclic compounds may be aromatic or non-aromatic, in the latter case, they may vary from being fully saturated to having varying numbers of multiple bonds between the ring atoms.
Based on the constitution of the rings, cyclic compounds are further divided into
- Homocyclic or carbocyclic compounds
- Heterocyclic compounds
Homocyclic compounds | Heterocyclic compounds |
Homocyclic compound are cyclic compound having atoms of the same element as ring members | Heterocyclic compounds are cyclic compounds having atoms of the different elements as ring members including carbon atoms. |
Ring contains atoms of the same element | Ring contains atoms of different elements |
Contain atoms of the same element bonded to each other forming a ring | Contains atoms of at least two different elements bonded to each other forming a ring |
E.g.: include benzene, cyclohexane, cyclohexanol etc. | E.g.: include pyran, azocine, thiocane etc. |
Chirality is a useful concept in physical and life sciences, especially when applied to a molecular level. It derives from the Greek work their meaning “hand” because, like hands, the molecules and objects in general are not super imposable with their mirror images. The term chiral was coined by Lord Kelvin in his Baltimore lectures, although its usage remained ignored for nearly one century, being rediscovered by the mid-1960s to define a geometrical model devoid of certain symmetry elements, with the exception of an axis of rotation. Chirality manifests itself in both molecules and crystals, and its origin lies clearly in molecular architecture. The analogy between crystals and molecules in this context was first caught by Pasteur, who realized that the nonidentity of the crystal (or molecule) with its mirror image was due to what he called dissymmetry (we would now say chirality). Although Pasteur himself conjectured on different structural arrangements, including tetrahedral, he was unaware of the tetrahedral asymmetric carbon postulated by van't Hoff and Le Bel in 1874. The two forms of a chiral molecule are called enantiomers and have identical physical and chemical properties, although they will rotate the plane of polarized light in opposite senses (i.e., optical activity). Two enantiomers will usually differ in reactivity in the presence of other chiral molecules, a fact of enormous significance in biology and drug discovery. Chiral and related words (e.g., homochiral, heterochiral) have engendered both confusion and ambiguity in the literature and, unfortunately, improper usage is not unusual. A brief, yet convenient, discussion illustrates the points. A single chiral molecule will either be right-handed or left-handed. The term chiral is insufficient, however, when applied to a compound or sample (i.e., a macroscopic collection of molecules), because it does not imply that we are dealing with molecules having the same sense of chirality. Introduction of homochiral or heterochiral labels are appropriate to this end. Moreover, homochiral means that the sample is made up of molecules with the same sense of chirality or handedness, which does not necessarily imply the existence of a single enantiomer. Clearly, one needs to know if the sample in question is actually racemic, non racemic, in which the chiral sample contains a certain excess of the major enantiomer. Finally, as one would have expected for a term restricted to molecules and objects, chiral and chirality should not be employed for dynamic transformations (e.g., chiral synthesis, chiral separation, chiral amplification and so on) despite their widespread use. The situation may even be more complex in crystals because a racemic crystal is not equivalent to a racemic compound in terms of molecular composition. In addition, the distinctive nature of stereoisomers in the solid state is crucial to understand the separation and resolution of enantiomers by crystallization.
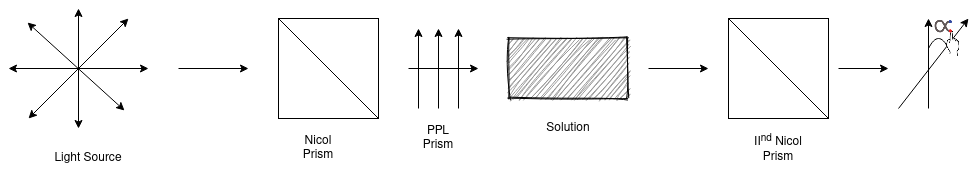
The instrument used for measuring optical rotation is called polarimeter. It consists of light source to nicol prism and in between a tube to hold solution of organic substance. The schematic representation of polarimeter is given as –
Isomerism:
The compound having same molecular formula but different physical and chemical properties due to the different arrangement of atoms or molecules are known as isomers and the whole phenomena is called as Isomerisation.
Structural Isomerism:
It is due to the difference in ways in which different atoms or groups are linked to one another within the molecule. In this type of isomerism the isomers have different structure without taking into consideration the arrangement of groups in space. These are further classified as:
Chain Isomerism:
It arises due to the different arrangement of C atoms that is straight or branch chain of C atoms. E.g.: n-butane & iso-butane have same molecular formula C4H10.
n-butane - straight chain
Iso-butane - branched chain
Position Isomerism:
It is due to the difference in position taken up by substituent atoms or groups in same C chain or due to different position of double or triple bond. E.g.: C3H7Cl shows 2 isomeric compound i.e.; n-propyl chloride and iso-propyl chloride.
Ring Chain Isomerism:
This type of isomerism arises due to the difference in type of linking of C-atom & isomer may have either open chain or closed chain structure.
Functional Isomerism:
It arises due to the difference in nature of functional group present in isomeric compound. E.g.: C2H6O
CH3-O-CH3 CH3-CH2-OH
Dimethyl ether ethanol
Compounds having identical structure but difference in their configuration i.e.; relative arrangement of atoms or groups within the molecule or different in space are called stereoisomers. The phenomena is called as the stereoisomerism. These compound are said to have different configuration.
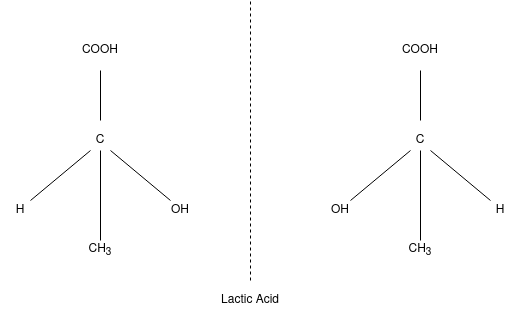
Lewis Pastues while studying crystallography of salt of tartaric acid. He observed that optical inactive Na, NH3, tartrate exists as mixture of 2 different type of crystals which mirror images of each other. These crystal which gives the mirror images of each other are called enantiomorphs & the phenomena are called as the enantiomorphism. Although the original mixture was optically inactive each type of crystal when dissolved in water were found to be optically active. The specific rotation of 2 solution were exactly equal but of opposite sign i.e.; 1 solution rotated the PPL to the right or clockwise while other to the left or anti clockwise to the same extent, 2 type of crystal or solution were identical in all other physical and chemical properties. Isomers which are non super imposable mirror images of each other are called enantiomers.
According to the Le-Bell and Van’t Hoff the 4 valencies of C atom are directed towards the 4 corners of a regular tetrahedron at the centre of which C atom lies. Consider a compound CLMNO in which L,M,N,O are 4 different group or atom attached to the C atom may be represented by 2 models:
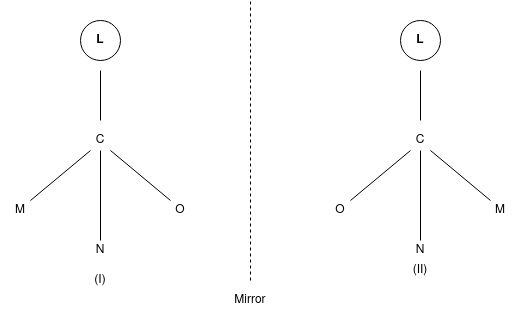
It is important to know that these 2 molecules can’t be super imposed on each other.
E.g.; Lactic acid and secondary butyl chloride exists as 2 optically active isomers which are enantiomers i.e.; mirror images of each other. Mirror images of 2 compounds are shown as:
Diastereomers are defined as compounds which have the same molecular formula and sequence of bonded elements but which are non super-imposable, non-mirror images.
Enantiomers and diastereomers commonly called stereoisomer’s fall under the broader concept of isomerism which always involves the comparison of at least two species. Stereoisomers are molecules of identical constitution but nevertheless different. Differences between diastereomers can be expressed in scalar terms, that is by differences in the distances of certain characteristic pairs of atoms. Compounds having identical structure but difference in their configuration i.e.; relative arrangement of atoms or groups within the molecule or different in space are called stereoisomers. The phenomena is called as the stereoisomerism. These compounds are said to have different configuration.
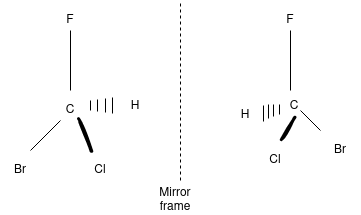
Carbon, when neutral, is bonded to four groups in a tetrahedral arrangement. What this means is that each group points as far away from the other in three-dimensional space as it can. Imagine carbon in the center of the tetrahedron shown below, with each of the four atoms that it is bonded to pointing in the direction of each corner. This describes the tetrahedral geometry of carbon. When a molecule contains a chiral carbon atom, there are four different groups attached to the carbon. Because of the way the groups orient themselves in space, the molecule can exist in one of two configurations at that carbon, called the absolute configuration. If the molecule has just one chiral carbon, these two configurations are called enantiomers. The two possible molecules are mirror images of each other, but they are non-superimposable. Take a peek at the two organic molecules below; they are mirror images, but if you tried to lay them on top of each other, the groups would never line up perfectly. This is what is meant by 'non-super imposable. This is a tough concept, so let's consider a real life example. Take the palms of your hands and face them toward each other. The thumbs line up, and so do your pinky fingers. The hands are mirror images. Now, try to put one hand on top of the other with he palms facing down. No matter how we position the hands, the thumbs will never line up. This is because they are non-superimposable.
You are more chiral than just your hands. In fact, 19 out of 20 of the amino acids that make up your body are chiral. The proteins that carry out your biological functions are made of these amino acids, so your proteins are chiral too. This means that they can interact with molecules containing chiral carbons distinctly. A protein may recognize one enantiomer of an organic molecule as a drug, and the enantiomer of that same organic molecule may have little to no effect.
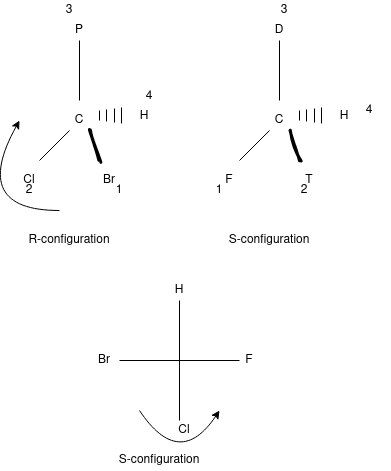
The spatial arrangement of the atoms of a chiral molecular group and its stereo chemical description.
R means Rectus while the S stands for the Sinister in regular term Rectus is orientation of molecule in clock wise direction while Sinister means the orientation of the molecule in anti clockwise direction. R-S configuration is used to give the identity to the elements attached to the chiral atoms.
The molecule must be arranged according to the priority order.
Priority order depends on the atomic number of the molecule. (the greater atomic number the greater is priority order and vice versa)
These isomers occur in the place of the restricted rotation of the molecule. Isomerism that arises out of the difference in the spatial arrangement of atoms or groups about the doubly bonded carbon atoms is called Geometrical Isomerism.
These isomers are not mirrored images of each other. Rotation about C=C is not possible under normal conditions, and hence the isomers are isolable.
If different atoms or groups are bonded to the 'C=C' bond in a molecule, more than one spatial arrangement is possible.
E.g.; 2 - butene exists in two isomeric forms.
The isomer in which similar groups lie on the same side is called 'Cis isomer' (I). The other in which similar group lie in opposite direction is called 'Trans isomer' (II). This isomerism is called 'Cis-Trans' isomerism. The two groups attached to the carbon atoms need not be the same, it may be different also,
This isomerism arises out of the hindrance to rotation about the C=C bond in such molecules.
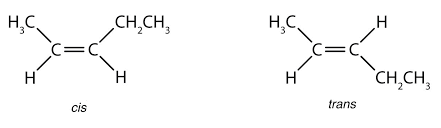
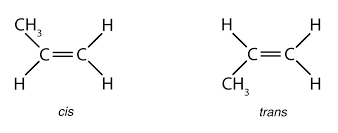

A mixture of equal parts of enantiomers is called a racemic modification. A racemic modification is optically inactive: When enantiomers are mixed together, the rotation caused by a molecule of one isomer is exactly cancelled by an equal and opposite rotation caused by a molecule of its enantiomer. The prefix ± is used to specify the racemic nature of the particular sample, as, for example, (±) -lactic acid or (±) -2-methyl-1-butanol. It is useful to compare a racemic modification with a compound whose molecules are super imposable on their mirror images, that is, with an achiral compound. They are both optically active.
Formation of Racemic Foundation:
By mixing the most obvious and trivial way of forming a racemic modification is by intimate mixing of exactly equal amounts of dextrorotatory (+) and laevorotatory (-) isomers. This method is related to an entropy of blending, since the racemic modification represents a lot of random state of affairs than the separate enantiomers.
By synthesis:
The synthesis of dissymmetric molecules, starting from either symmetric molecule of a racemic modification and using no optically active reagent or catalyst and no asymmetric physical influence always products a racemic modification the two enantiometric type of product molecules. The two common ways of production an asymmetric carbon in mole by displacement and by addition. The method is exemplified by addition of hydrogen cyanide to acetaldehyde to give lactonitrile. Here, approach from either side of carbon is equally facile, and therefore, equal number of mole of two enantiometric forms of lactonitrile, CH3 CHOHCN results.
By chemical transformation:
It is possible to change a dissymmetric mole to its enantiomeric form without ever breaking any of the bonds leading to the asymmetric atom.
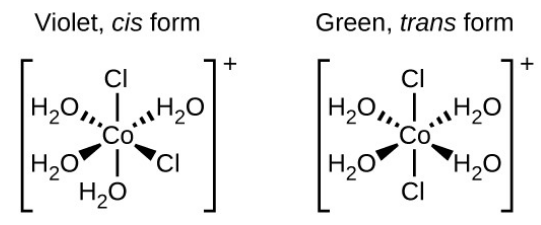
Isomers are different chemical species that have the same chemical formula. Transition metals often form geometric isomers, in which the same atoms are connected through the same types of bonds but with difference in their orientation in space. Coordination complexes with two different ligands in the cis and trans position from a ligand of interest form isomers. E.g.: the octahedral [Co(NH3)4Cl2]+ ion has two isomers. In the cis-configuration the two chloride ligand are adjacent to each other. The other isomer, the trans configuration has the two chloride ligands directly across from one another.
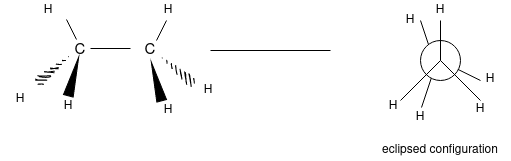
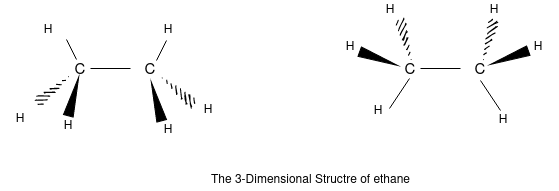
The 3 dimensional structure of ethane (C2H6):
Ethane Confirmations:
Ethane molecule consists of 7 sigma bond on the rotation there are no changes noticed in the 6 carbon hydrogen molecule as because the hydrogen are essentially spherical.
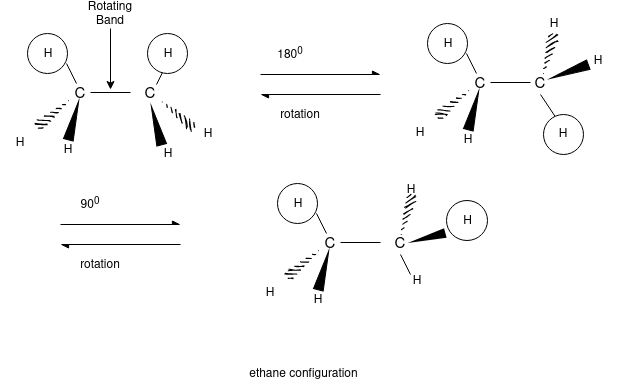
In order to better visualize these different conformations, it is convenient to use a drawing convention called the Newman projection. In a Newman projection, we look lengthwise down a specific bond of interest – in this case, the carbon-carbon bond in ethane.
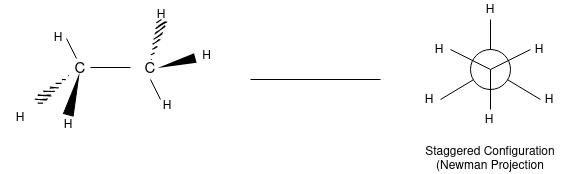
The hydrogen and carbon atom depicted above shows the 120o bond angles. While the lowest conformation of ethane is called as the Staggered Conformation in which all of the C-H bonds on the front carbon are positioned at dihedral angles of 60°relative to the C-H bonds on the back carbon. In this conformation, the distance between the bonds (and the electrons in them) is maximized.