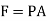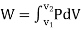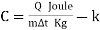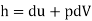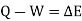Unit - 1
Review of Fundamental Concepts and Definitions
Basic concepts of thermodynamics
Thermodynamics deals with Energy interactions. Energy interactions is possible by two methods namely Work transfer & Heat transfer.
When a substance changes from one condition to another in a process, energy transformation may occur. Common processes are heating, cooling, expansion, compression.
Working Substance or Medium
Thermodynamic process requires a carrier that would act as mode of transport of energy from or into the system. Such a medium is called as working substance or working medium.
For example, petrol is the working medium in SI engines whereas diesel is the working medium in CI engines.
System
Thermodynamic system is the region or quantity of matter which is under observation for thermodynamic changes. The quantity of matter can be gas, solid, liquid.
Everything which is external to the system is termed as surroundings / environment.
A system boundary separates system and surrounding. It can be both fixed and movable. A closed surface surrounding in a system through which energy & mass may enter or leave the system is called as Boundary.
System & surrounding when combined form Universe.
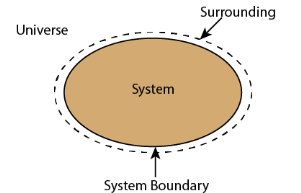
Figure. 1
- An isolated system cannot exchange either mass or energy with the surroundings.
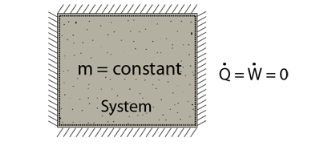
Figure. 2
- A closed system exchanges energy with the surrounding but mass transfer is not possible.
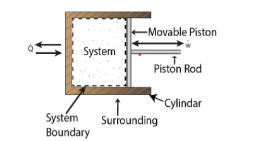
Figure. 3
- An open system can exchange both mass and energy with the surroundings.
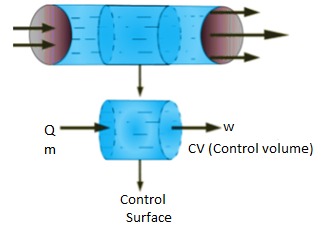
Figure. 4
There are two points of view from which the behavior of matter can be studied: the macroscopic and the microscopic.
- Macroscopic
The macroscopic approach considers a limited number of matters while ignoring events occurring at the molecular level. Macroscopic thermodynamics is only concerned with the effect of many molecules acting together, and these effects are perceptible to human senses. For example, the macroscopic quantity, pressure, is the average rate of change of momentum due to all the molecular collision made on a unit area. The effect of pressure can be felt. The macroscopic point of view is not concerned with the action of individual molecule, and the force on a given unit area can be measured by using e.g., a pressure gauge. These macroscopic observations are completely independent of assumptions regarding the nature of matter. All the results of classical or macroscopic thermodynamics can, however, be derived from the microscopic and statistical study of matter.
- Microscopic
From the microscopic point of view matter is composed of myriads of molecules. If it is a gas, each molecule at a given instant has a certain position, velocity, and energy, and for each molecule these changes very frequently because of collisions. In microscopic or statistical thermodynamics, the behavior of a gas is described by summarizing the behavior of each molecule..
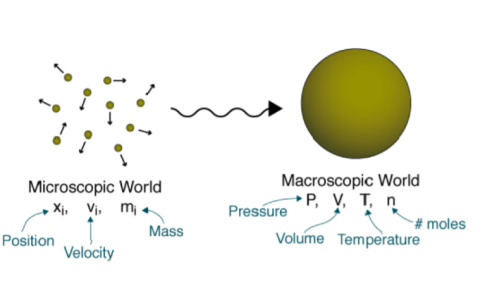
Figure. 5
“Continuum is defined as continuous distribution of mass within the matter or system with no empty space “
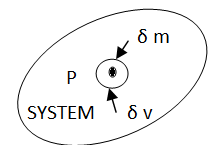
From the macroscopic viewpoint, we are always concerned with volume which is very large compared to molecular dimensions. Even a very small volume of a system is assumed to contain many molecules so that statically averaging is meaningful and a property value can be assigned to it. Disregarding the behavior of individual molecules, matter is here treated as continuous.
Let us consider a mass ∂m in a volume ∂V surrounded the point P (Fig 1). The ratio ∂m/∂V is the average mass density of the system within the volume ∂V. We suppose that at a first ∂V is rather large and is subsequently shrunk about the point P. If we plot m/ v against v, we can see that the average density fluctuates significantly with time as molecules pass into and out of the volume in random motion, making it impossible to speak of a definite value of ∂m/∂V. The smallest volume which may be regarded as continuous is ∂V’. As a result, the density of the system at a given
Point is defined as 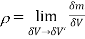 Definition of the macroscopic property, density
Definition of the macroscopic property, density
Similarly, the fluid velocity at a point P is defined as the instantaneous velocity of the smallest continuous volume ∂V'.
When the mean free path of the molecules approaches the order of magnitude of the vessel's dimensions, as in highly rarefied gases encountered in high vacuum technology, rocket flights at high altitudes, and electron tubes, the concept of continuum loses validity. However, in most engineering applications, the assumption of a continuum is valid and useful, and it goes hand in hand with the macroscopic point of view.
If the state of a system doesn’t have tendency to be able to change spontaneously then it is called to be in thermodynamic equilibrium.
i.e., The properties of which do not change with time and that can be changed to another condition only at the expense of effects on other systems.
Figure. 6
The 3 conditions must be satisfied to in in thermal equilibrium:
- Mechanical equilibrium
- Chemical equilibrium
- Thermal equilibrium
The precise state of a substance is referred to as its state variables, which determine the state of a substance are referred to as its properties or parameters.
Internal energy, pressure, temperature, and volume Enthalpy, entropy, and heat are properties that can be used to precisely place the exact condition of a substance. A minimum of two of these properties are required to pinpoint the state of any substance.
Properties are classified as Extensive & Intensive properties if a physical property of a system which does not depend on the system size or the amount of material in the system is known as Intensive properties. Extensive properties, on the other hand, are those that are dependent on the system size or the amount of material in the system.
Process and Cycle
Cycle and process When some property of a substance undergoes changes, it is said to have undergone a process. The process is named after its specifications, such as constant pressure process, constant volume process, and so on.
A cycle is a set of processes that results in the same initial and final states of a system. A thermodynamic cycle is also known as a cyclic process operation. Energy can be defined as a body's ability to perform work.
Energy:
Energy may be defined as the capacity of a body to perform work It comes in a variety of forms and can be converted from one to another. Energy is a path function, which means that it is determined by the process's path rather than the end states the process and not on the end states.
Exact differentials:
Path functions are properties that are expressed at a specific state of the system and are not at all dependent on the path taken by the system. Examples include pressure, temperature, volume, and so on.
Exact differentials are the derivatives of some other functions as the name suggests.
These are used to express change in point functions. Examples are pressure change dP, volume change dV, etc.
Inexact differentials:
Path functions are properties that depend on the path taken by the system rather than the state of the system. Work heat inexact differentials are useful for expressing very small quantities in these path functions.
When a process is carried out infinity slowly, it is referred to as a quasistatic process.
When a process proceeds in such a way that the system always remains infinitesimally close to an equilibrium state: quasi-static or quasi-equilibrium process the process proceeds slowly enough to allow the system to adjust itself internally so that properties in one part of the system do not change any faster than those in other parts of the system.
It is a thermodynamic process that occurs at an infinitesimal rate. Every state that the quasi-static process passes through tends to equilibrium. The terms quasi and static refer to thermodynamic properties that remain constant over time. A reversible process is a quasistatic process.
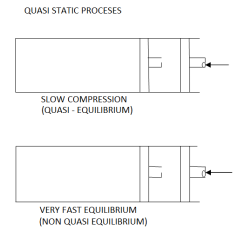
Figure. 7
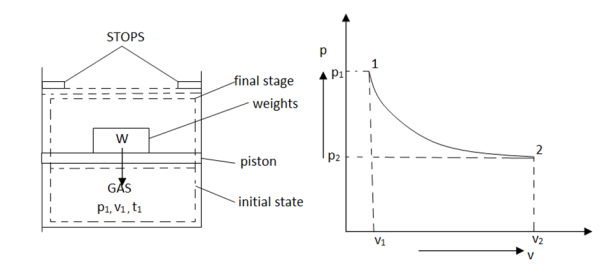
Figure. 8
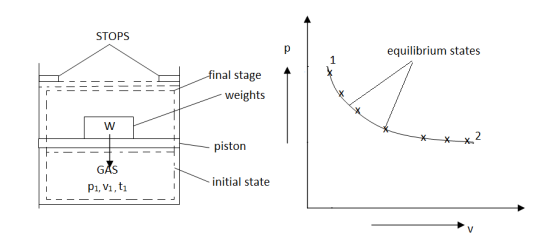
Figure. 9
Infinite slowness is characteristic feature of quasi static process.
If the gradient is present in system properties, then the process cannot be quasi static for a finite time. However, if the process's time is infinite, the process can be quasi-static, even if there is some gradient.
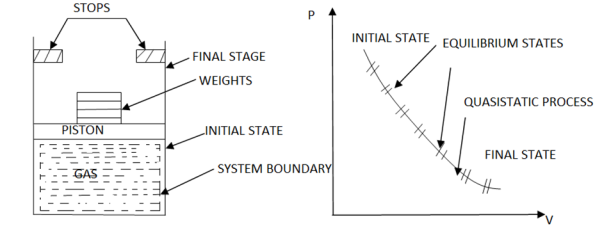
Figure. 10
As in figure the piston cylinder assembly is shown and the gas is in equilibrium by the constant pressure (weight) applied on piston and its state can be represented by thermodynamic properties p1, v1, t1.
If the weights are suddenly removed, then the gas will force the piston upwards until the stops or another equilibrium state.
The final equilibrium state can be represented by thermodynamic properties p2, v2, t2.
Now the in between states are not in equilibrium as the spontaneous process occurs. If the weights are now divided into infinite parts and it is taken out one by one so that the process moves on through the successive equilibrium states.
This is a quasi-static process example.
If the system's initial state cannot be restored without causing changes to the system or its surroundings, the process is known as irreversible. The following conditions must be met:
• It has to be a quasi-static process.
• Friction, viscosity, and other such properties should not be present in the system.
The concept of reversible process is speculative.
• External: Physical factors such as friction, resistance, viscosity, surface tension, finite temperature difference, and others cause irreversibility.
Because the energy lost cannot be recovered, the process cannot be reversed without the application of external work.
• Internal: Irreversibility is caused by the working fluid's properties in a process such as throttling or free expansion.
The gas expands due to its internal energy. It cannot contract and reverse the process on its own.
• Chemical: Internal chemical properties such as structure, bonds, and so on cause irreversibility.
When a chemical reaction occurs as a result of heat absorption or rejection, it cannot reverse spontaneously.
Work or Displacement work
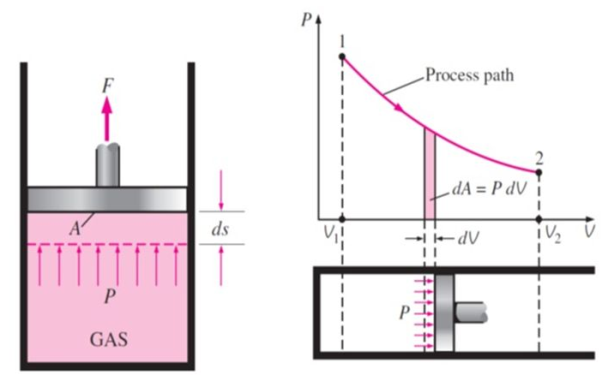
Figure. 11
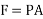
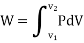
This PdV can be performed only on a quasi-static path.
Work done is measured in Joule (J) or Newton meter (Nm). Rate at which work is done is called as power (Joule/second) or Watt.
Specific heat & latent heat (C)
The amount of heat required to raise a unit mass of the substance through a unit rise in temperature.
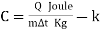
Heat Capacity (C /Cp/Cv)
The product of mass & specific heat is called the heat capacity.
Latent heat
The latent heat is the amount of heat transfer required to cause phase change in unit mass of a substance is a function of temperature.
- Latent heat of fusion
- Latent heat of vaporization
- Latent heat of sublimation
* Heat transfer is a path function
Work (w): In thermodynamics, work (w) is the energy transferred by a system to its surroundings. Kinetic energy, potential energy, and internal energy are all types of energy that exist as system properties. Work is a type of energy, but it is energy in motion. A system does not contain work; work is a process performed by or on a system. In general, work in mechanical systems is defined as the action of a force on an object over a distance.
Work performed by a system on its surroundings is regarded as positive, whereas work performed on the system by its surroundings is regarded as negative.
We know that
Work = force x displacement
But in case of thermodynamics works we take force is the product of a pressure p at the boundary and the area A of the moving boundary therefore
DW = pAdl
Where dW is the work done during an infinite displacement dl of the boundary
Since Adl = dv = change in volume
DW = pdv
The conditions under which the above expression is valid are
The system is closed
There are no viscous effects within the system.
The pressure and other properties have the same value on all boundaries of the system
The effects due to gravity electricity or magnetism are negligible
Heat (Q): Heat Q is a type of energy that is transferred across a boundary due to a temperature difference. Heat given by a system to its surroundings is regarded as negative, whereas heat rejected by the surroundings on the system is regarded as positive.
Key Takeaway:
Ideal gas follows all the gas laws that are stated below:
- Boyle's law: (at T = const)
P ∝ 1/ V or PV = k
2. Charles' law: (at P = const)
V ∝ T or V/T = k
3. Gay-Lussac's law: (at V = const)
P ∝ T or P/T = k
4. Avogadro's law: Avogadro's law states that, "equal volumes of all gases, at the same temperature and pressure, have the same number of molecules”
V ∝ n or V/n = k
Where, n is the amount of substance of the gas (moles).
An ideal gas is a theoretical gas made up of a collection of randomly moving point particles that only interact via elastic collisions. The ideal gas concept is useful because it obeys the ideal gas law, which is a simplified equation of state, and is amenable to statistical mechanics analysis.
Most real gases behave qualitatively like ideal gases under normal ambient conditions such as standard temperature and pressure. In general, deviation from an ideal gas tends to decrease with higher temperature and lower density, as the work performed by intermolecular forces becomes less significant when compared to the kinetic energy of the particles, and the size of the molecules becomes less significant when compared to the size of the molecules.
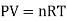
An ideal gas follows this equation.
Equation Table for an Ideal Gas
S.No | Name of the process | Parameter Held Constant | Remarks |
1 | Constant pressure (Isobaric) | p=constant | v=(mR/P)T |
2 | Constant volume(isochoric) process | v=constant | p=(mR/v)T |
3 | Constant temperature (isothermal) process | T=constant | PV=constant |
4 | Polytropic process | n | 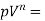 |
5 | Adiabatic process | No beat flow across the system boundary | 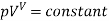 |
6 | Isoenthapic process | h=constant | h=constant |
Real gas effects, as opposed to Perfect or Ideal Gas effects, refer to an assumption base that takes the following into account:
•Compressibility effects
•Variable heat capacity
•Van der Waals forces
•Non-equilibrium thermodynamic effects
•Issues with molecular dissociation and elementary reactions with variable composition.
For most applications, such a thorough examination is "overkill," and the ideal gas approximation is used. Real-gas models must be used near the condensation point of gases, near the critical point, at extremely high pressures, and in a variety of other unusual situations.
In certain gases, the compressibility factor Z is a function of P & T. On the coordinates of P & Z when the plot of constant Temp is made.
From this plot P & Z can be found out, on the other hand V is calculated from 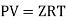
For each substance there is a compressibility factor chart. The shapes of vapor dome and of the constant temperature lines on the P-V planes are similar for all substances. But remember scales are different. This is exploited by using dimensionless properties also known as reduced properties.
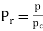 ,
, 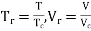
r = reduced state & c = Critical state
The relation between this reduced state is known as Law of corresponding states.
Daltons Law of partial pressure
In a homogeneous mixture of inert gases, having temperature T, Pressure P, Volume V. Where n = number of moles
Then,
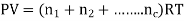
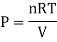
So, P1 + P2+ P3+………+Pc = P
- Electrical Work: The work required to move a unit charge once around the entire circuit is defined as electrical work.
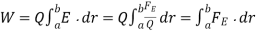
Here, Q = charge on the particle.
E = strength of electric field,
FE = Coulomb force
r = displacement of particle under the action of the Coulomb force.
- Magnetic Work:
Magnetic work is the work done by a unit magnetic field in moving a unit charge. It is provided by,
W = F dR
Where, F = force of magnetic field = qv X B
Dr = distance of movement of charge.
- Gravitational Work:
Gravitational work is the work done in moving someone against the force of gravity.
Force due to gravity = F = mg
Gravitational work = F x h = mgh
Where, h = lift of the particle against the force of gravity.
- Spring Work:
When a force is applied to a spring, it will either compress or expand depending on the direction of the force.
This displacement requires some effort from the spring.
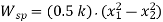
Where, k = stiffness constant of the spring,
x1 = initial distance of the spring from datum
x2 = final distance of the spring from the datum
- Shaft Work:
Work is done on a shaft when it is rotated by an external force.
It is given by,
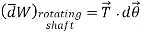
Where, T= torque acting on the shaft
dθ = angular displacement in radians.
Temperature:
A substance's temperature is defined as the degree of hotness or coldness of a body. We know that energy in the universe flows from higher to lower levels. As a result, heat flows from a hotter body to a colder body.
Temperature is not a measure of the quantity of energy in the body; rather, it indicates the level of internal energy in the body.
The absolute temperature is defined as the temperature measured above absolute zero. The capital letter T is used to represent absolute temperatures.
Thermal Equilibrium:
Thermal equilibrium is the absence of heat transfer between two bodies in contact or between a system and its surroundings.
Zeroth Law of Thermodynamics:
If Body 1 is in thermal equilibrium with Body 3 and Body 2 is in thermal equilibrium with Body 3, it implies that Body 1 is also in thermal equilibrium with Body.
Key Takeaway:
- Temperature is defined as the degree of hotness or coldness of a body
- Thermal equilibrium is the absence of heat transfer between two bodies in contact or between a system and its surroundings.
- If Body 1 is in thermal equilibrium with Body 3 and Body 2 is in thermal equilibrium with Body 3, it implies that Body 1 is also in thermal equilibrium with Body.
Temperature Scales:
There are mainly four types of temperature scales.
- Fahrenheit Scale: It is widely used in the United States. Daniel Gabriel Fahrenheit, a German scientist, founded the company. It is capable of expressing negative temperatures. The following are the key points on this scale:
- Freezing point of water: 32° F
- Boiling point of water: 212° F
- Coldest possible temperature: -459.67° F (absolute zero)
b. Celsius Scale: The most widely used system and scale in the world. It is also known as the Centigrade scale because it divides the difference between the freezing and boiling points of water into 100 divisions. It is also capable of expressing negative temperatures. The following are the key points on this scale:
- Freezing point of water: 0° C
- Boiling point of water: 100° C
- Coldest possible temperature: -273.15° C (absolute zero)
- Fahrenheit to Celsius formula Celsius = (Fahrenheit - 32) / 1.8
c. Kelvin Scale: It is derived from the Celsius scale. It was intended to set the absolute zero temperature to 0 degrees Celsius. The degree scale is not used in this scale. The following are the key points on this scale:
- Freezing point of water: 273.15 K
- Boiling point of water: 373.15 K
- Coldest possible temperature: 0 K (absolute zero)
- You can convert from Celsius to Kelvin by adding 273.15 to Celsius temperature.
d. Rankine Scale:It is derived from the Fahrenheit scale. It was intended to set the absolute zero temperature to 0 degrees Celsius. The following are the key points on this scale
- Freezing point of water: 491.67 R
- Boiling point of water: 671.67 R
- Coldest possible temperature: 0 R (absolute zero)
- You can convert from Fahrenheit to Rankine by adding 459.67 to Fahrenheit temperature.
If a system undergoes a change of state during which both heat transfer and work transfer are involved, the net energy transfer will be stored or accumulated within the system.
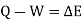
It can also Know as conservation of Energy which is stated as “Energy Can neither be created nor destroyed, but only gets transferred from one form into another”
Energy in storage is neither heat nor work & is given the name internal energy (E).
Energy has a definite value at every state of a system & therefore a property of system. Also, cyclic integral of any property is zero, because the final state is identical with the initial state.
Different form of stored energy
Kinetic energy = KE
Potential energy = PE
KE + PE = Bulk energy
Molecular energy = Internal energy (U)
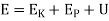
Enthalpy (h)
Defined as the sum of the system's internal energy and the product of its pressure and volume
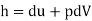
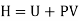
Key Takeaway
First law of thermodynamics applied to control Volume **
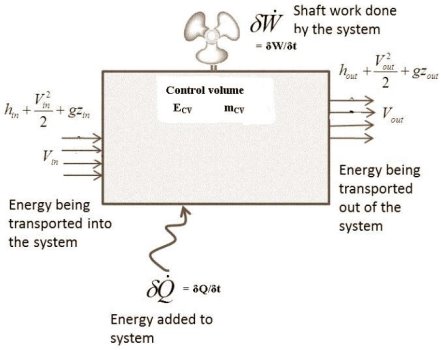
Figure. 12
Consider the control volume with an energy inflow, as shown in the diagram above.
As shown in the figure, the system is producing some shaft work.
The fluid in the system is constantly flowing in and out.
The rate of heat energy addition to the control volume = δQ/δt
The rate of work energy providing by control volume to the surrounding = δW/δt.
The mass flow rate entering to the control volume = dm1/dt
The mass flow rate leaving the control volume =dm2/dt
Rate of change of mass in control volume = dmCV/dt
According to principle of conservation of mass, we will have following equation
(Mass flow rate entering to the system) - (Mass flow rate leaving the system) = Rate of change of mass in control volume
(dm1/dt) - (dm2/dt) = dmCV/dt
E = u + pv + V2/2 + gz
E = h + V2/2 + gz
Ein = hin + V2in/2 + gzin = h1 + V21/2 + gz1
E1 = h1 + V21/2 + gz1
Eout = hout + V2out/2 + gzout = h2 + V22/2 + gz2
E2 = h2 + V22/2 + gz2
Steady Flow System -
Defined as the system in which the mass flow rate into the system is equal to mass flow rate out of the system.
Mass flow rate in m1 = Mass flow rate out, m2 = constant = m
Consider the flow of fluid through a pipe of cross-sectional area A,
Specific volume V, at Velocity C.
Volume flow rate = A (m2) × C (m/s)
Mass flow rate m (kg/s) = Volume flow rate / Specific volume
m =AC/V = ρAC= Continuity Equation
Notations | At Inlet | At exit | Units |
Pressure |  |  | m3/kg |
Specific volume | 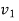 | 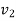 | J/kg |
Velocity | 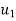 | 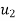 | m/s |
Elevation from arbitrary datum | 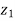 | 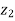 | m |
Mass flow rate | 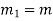 | 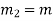 | Kg/s |
Cross-section of stream | 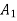 | 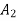 | m2 |
Specific enthalpy | 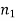 | 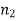 | J/kg |
Heat transfer rate to the system = Q [ J/s]
Work transfer rate from the system = W [ J/s]
Assumptions-
1. At no time does mass accumulate or decrease in the control volume.
2. The rate of mass transfer at the inlet and exit is the same.
3. The state and energy of the fluid at the inlet, exit, and control volume do not change over time.
4. Heat and work transfers across the control volume occur at a constant rate.
S.F.E.E. (Steady flow equation) on unit time basis -
Total energy flow rate into the control = total energy flow rate out of control volume.
Q+ m [Internal energy + Flow work + KE + PE ]1 = W +m [Internal energy + Flow work + KE + PE]2
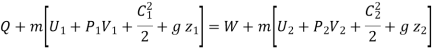
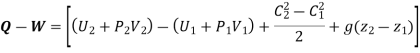
Enthalpy –h=U+PV
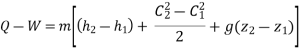
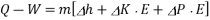
Boiler - A boiler is a device that produces steam at a constant pressure.
Heat is supplied to the boiler drum from outside by the combustion of fuel in the presence of air. Flue gases are the by products of combustion.
For a boiler - W =0, ΔK.E.=0 and Δ P.E.=0
On using these conditions S.F.E. Equation,
Q = m (h2 - h1)
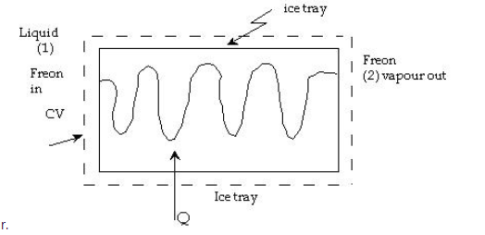
Heat Exchanger (Condenser/Evaporator) -
A condenser is a device that allows for constant-pressure steam condensation.
For a condenser - W =0, ΔK.E.=0 and Δ P.E.=0
On using these conditions S.F.E. Equation,
Q = m (h1 – h2)
The velocities are small from SFEE
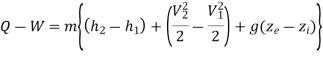
Where 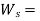 No external work done
No external work done
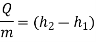
Figure. 12
Nozzle: In the case of a nozzle, the flow of fluid is accelerated as the enthalpy of the fluid decreases and the pressure drops at the same time. This is commonly used to convert a portion of the energy in steam into kinetic energy of the steam supplied to the turbine.
For this system, ∆PE=0, W=0, Q=0
Applying the energy equation to the system,
h1+(C12/2) =h2+(C22/2) h1+(C12/2) =h2+(C22/2)
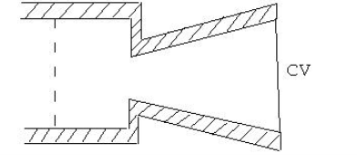
Figure. 13
While for high velocity its convergent divergent. Writing SFEE:
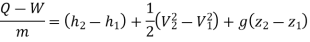
Where Q=adiabatic
W= No external work being done
Here 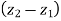 can be ignored
can be ignored
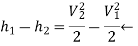
Ie the increase in k.e. Is equal to the decrease in enthalpy.
Turbine: In a steam or gas turbine, steam or gas is passed through the turbine, with a portion of its energy converted into work in the turbine. The turbine's output powers a generator, which generates electricity. The pressure or temperature of the steam or gas as it exits the turbine is lower.
Applying energy equation to the system,
Here, Z1=Z2
h1+(C12/2) −Q=h2+(C22/2) +W
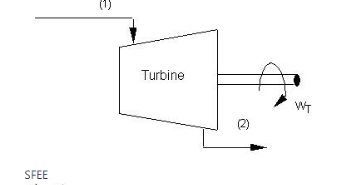
Figure. 14
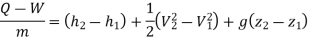
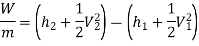
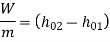
The quantity
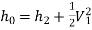 is known as the stagnation enthalpy
is known as the stagnation enthalpy
Throttling Device: Throttling occurs when a high-pressure fluid passes through a narrow or restricted region with no heat transfer to the surrounding area. This is an irreversible process. During throttling, pressure falls while volume rises. Enthalpy does not change.
SFEE can be written between (2) and(!) if we choose (@) at a position where outgoing stream is fairly uniform
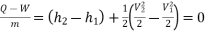
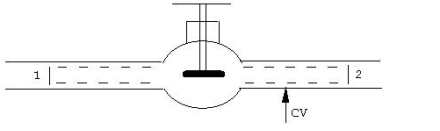
Figure.15
Here Q is dabatic and 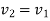
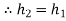
Steady Flow | Unsteady Flow |
|
|
Boiler | Passage of a Flood Wave |
Condenser | Operation of Irrigation and Power Canals |
Nozzle | Tidal Effects |
Turbine | Junctions |
Compressor | Measures to Control Flood |
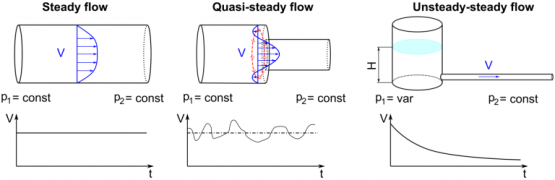
Figure. 16
1. The first law does not specify the maximum amount of energy that the thermodynamic system can convert to work.
2. While the first law explains the equivalence of work and heat transfers, it does not specify the conditions that allow for this interconversion.
It does not set any limit on direction of heat flow
It is a fictitious concept. This would be a violation of thermodynamics' first law. It generates energy without any input, and as we all know, according to the law of energy conservation, energy cannot be created or destroyed, but can only be converted from one form of energy to another.
As a result, a machine that does not obey the first law of thermodynamics or the law of conservation of energy is referred to as a perpetual motion machine of the first kind, or PMM1. This means that there can be no machine that continuously supplies mechanical work without some other form of energy disappearing at the same time.
When a fluid flows through a control volume, its thermodynamic properties can change along space coordinates as well as over time. If the rates of flow of mass and energy through the control surface change over time, so will the mass and energy within the control volume.
The term' steady flow' refers to the constant rates of mass and energy flow across the control surface.
At the steady state of a system, any thermodynamic property has a fixed value at a specific location and does not change over time.
Thermodynamic properties vary along space coordinates but not with time. The term' steady state' refers to a state that is constant or invariant over time.
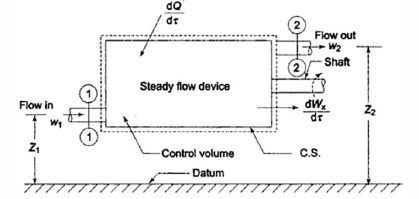
Figure. 17
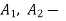 crosss-section of stream,
crosss-section of stream, 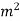
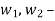 mass flow rate, kg/s
mass flow rate, kg/s
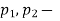 pressure, absolute
pressure, absolute 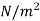
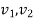 - specific volume,
- specific volume, 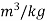
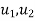 - specific internal energy, J/kg
- specific internal energy, J/kg
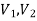 - velocity m/s
- velocity m/s
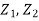 - elevation above an arbitrary datum m
- elevation above an arbitrary datum m
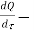 net rate of heat transfer through the control surface, J/s
net rate of heat transfer through the control surface, J/s
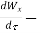 net rate of work transfer through the control surface J/s
net rate of work transfer through the control surface J/s
Exclusive of work done at sections 1 and 2 in transferring the fluid through the control surface
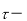 time,s.
time,s.
Mass Balance
According to the law of conservation of mass, if no mass accumulates within the control volume, the mass flow rate entering must equal the mass flow rate leaving.
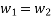
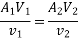
This is referred to as the equation of continuity.
Key Takeaway:
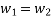
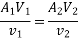
Work transfer in a flow process can be of two types: external work and flow work. The term "external work" refers to all work transfer across the control surface that is not caused by normal fluid forces. Shear (shaft or stirring) work and electrical work are the only types of external work that matter in engineering thermodynamics. Only external work, Wx, takes the form of shaft work. The flow work is the displacement work done by a fluid of mass dm1 at the inlet section I and a fluid of mass d2 at the exit section 2, which are (-p1 v1dm1) and (+p2v2dmi) respectively. As a result, total work transfer is given by
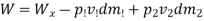
In the rate form 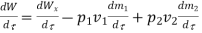
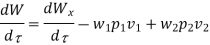
Because there is no energy accumulation, the total rate of flow of all energy streams entering the control volume must equal the total rate of flow of all energy streams leaving the control volume. This can be expressed mathematically as follows.
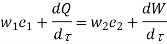
Substituting for 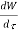 from eq(5.4)
from eq(5.4)
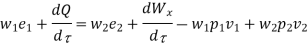
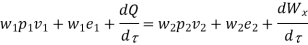
Where e1 and e2 denote the energy carried into or out of the control volume per unit mass of fluid.
The specific energy e is given by
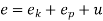
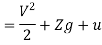
Substituting the expression for e
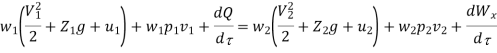
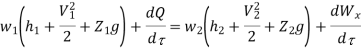
Where 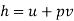
And since 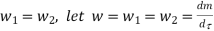
Dividing by 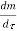
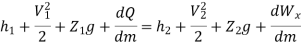
Or it can also be written as
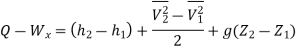
In differential form it is written as
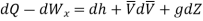
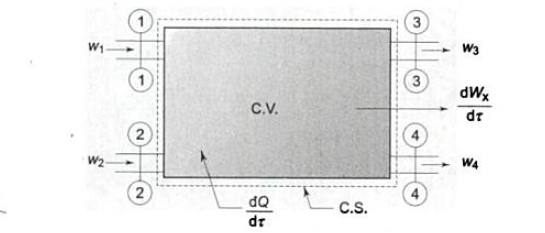
Figure. 18
Mass balance
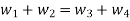
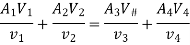
Energy balance
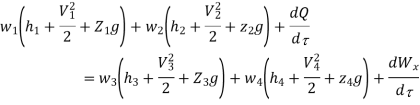
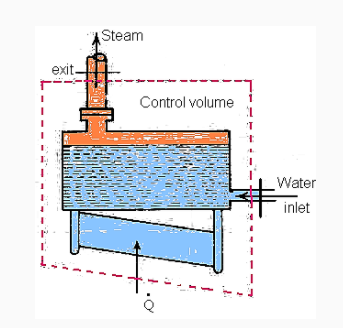
Figure. 19
(a) Velocity change between inlet and exit point is not appreciable and may be neglected 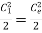
(b) Elevation change between inlet and exit point may be neglected
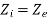
(c) There is no shaft work
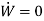
The steady flow energy equation is
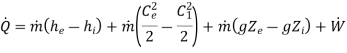
By applying the feautres of steam boiler, the above equation reduces to
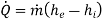
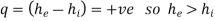
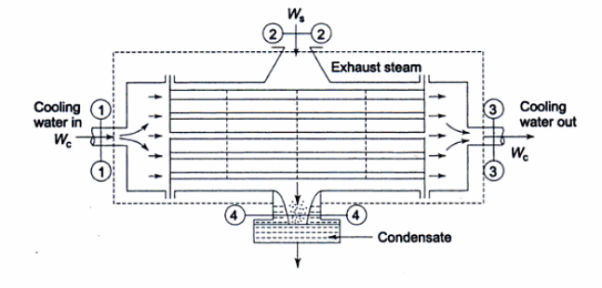
Figure 20
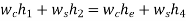
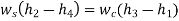
There is no external work done, and energy exchange in the form of heat is confined only between the two fluids, i.e., there is no external heat interaction or heat loss.
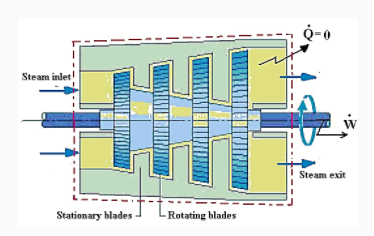
Figure 21
The steady flow energy equation is
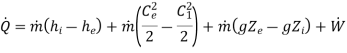
By applying the features of turbine, the above equation reduces to
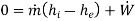
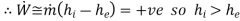
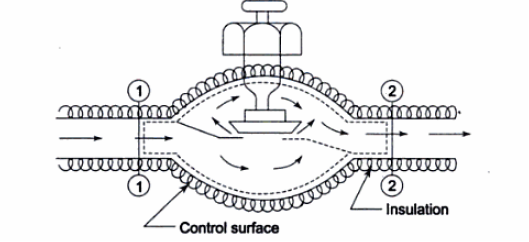
Figure. 22
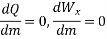
P.E. Changes are minor and are largely ignored. As a result, the S.F.E.E. Drops to
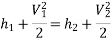
When throttling, the pipe velocities are frequently so low that the K.E. Terms are also negligible. So
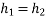
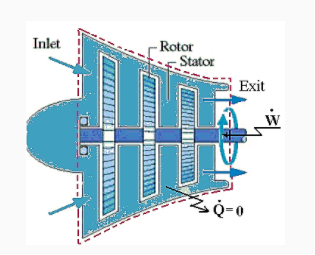
Figure 23
A compressor is a device that uses shaft work input to increase the pressure of a gas passing through it.
Pump: A pump is similar to a compressor, but it handles liquids.
Fan: A fan moves air with a slight pressure rise.
Usually 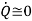 unless intentionally cooled, P.E≅0 and K.E small for compressors.
unless intentionally cooled, P.E≅0 and K.E small for compressors.
For the steady state steady flow process
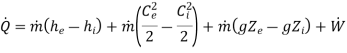
By applying the features of compressors/pumps/fans, the above equation reduces to
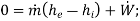
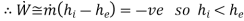
Note: for Reciprocating compressor 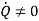
References:
- Basic and Applied Thermodynamics by PK Nag, MCGRAW HILL INDIA.
- Thermodynamics for Engineers by Kroos & Potter, Cengage Learning.
- Thermodynamics by Shavit and Gutfinger, CRC Press.
- Thermodynamics- An Engineering Approach by Cengel, MCGRAW HILL INDIA.
- Basic Engineering Thermodynamics, Joel, Pearson.